
An element has a body-centered cubic (bcc) structure with a cell edge of 288pm. The density...... - YouTube

Unit Cell Chemistry, Atomic Radius, Density & Edge Length Calculations, Close Packed Structures - YouTube

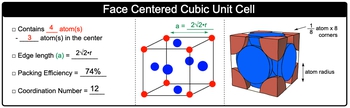
Naturally occurring gold crystallises in face centred cubic structure and has a density of 19.3 g cm ^-3 . Find atomic radius of gold. (Au = 197 g mol ^-1 )


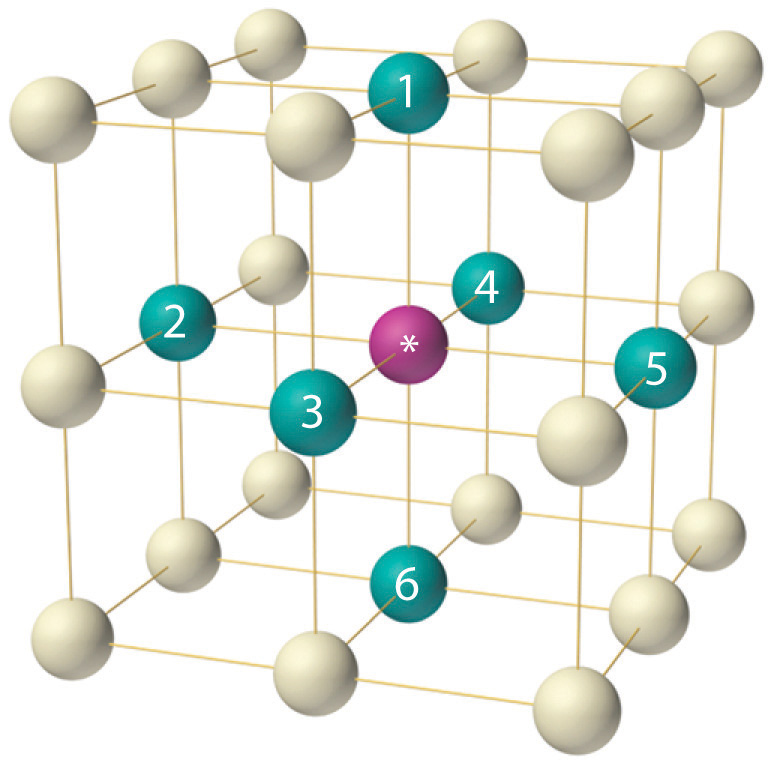


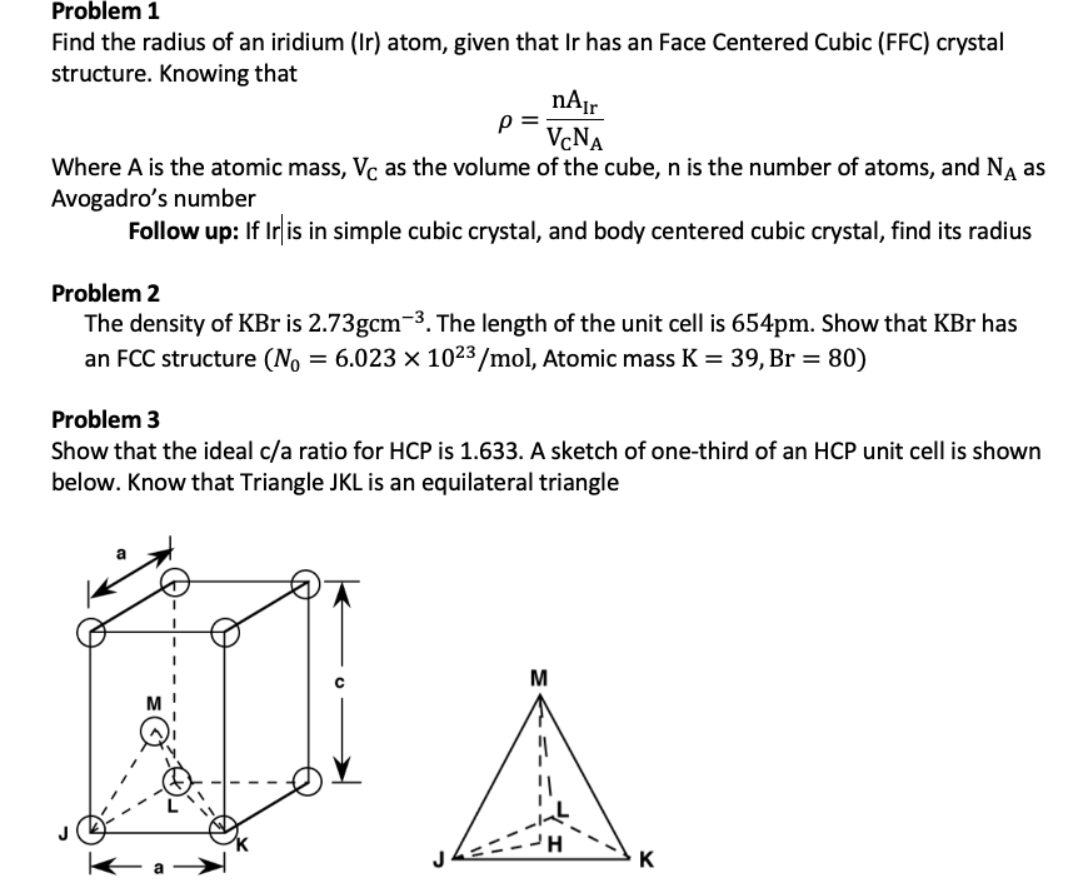
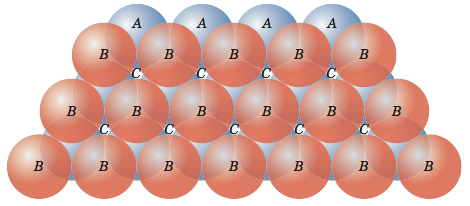
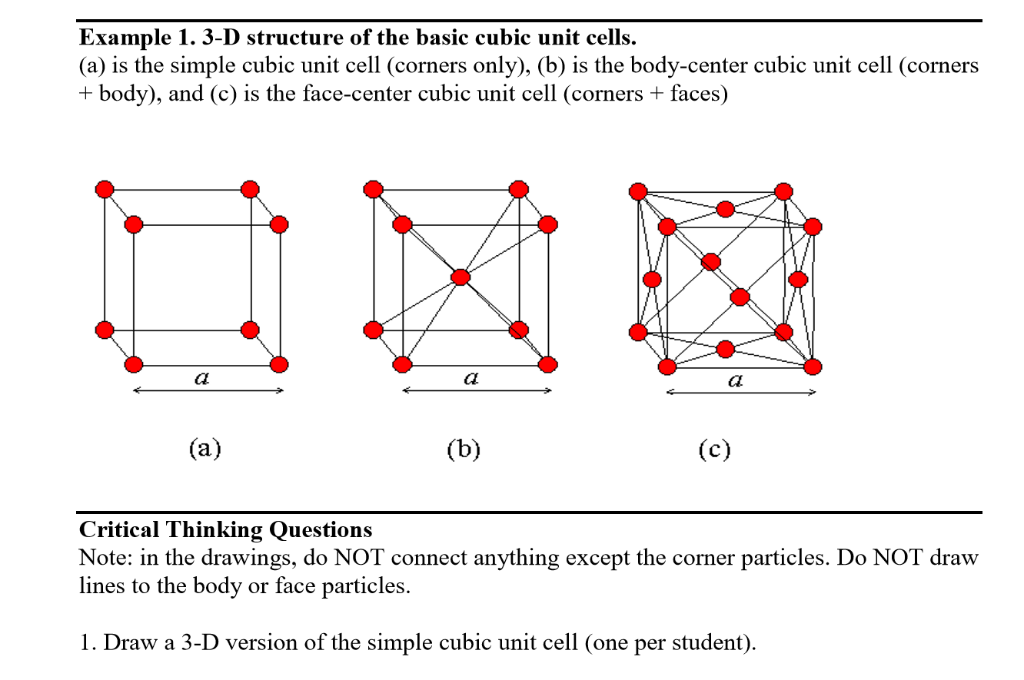






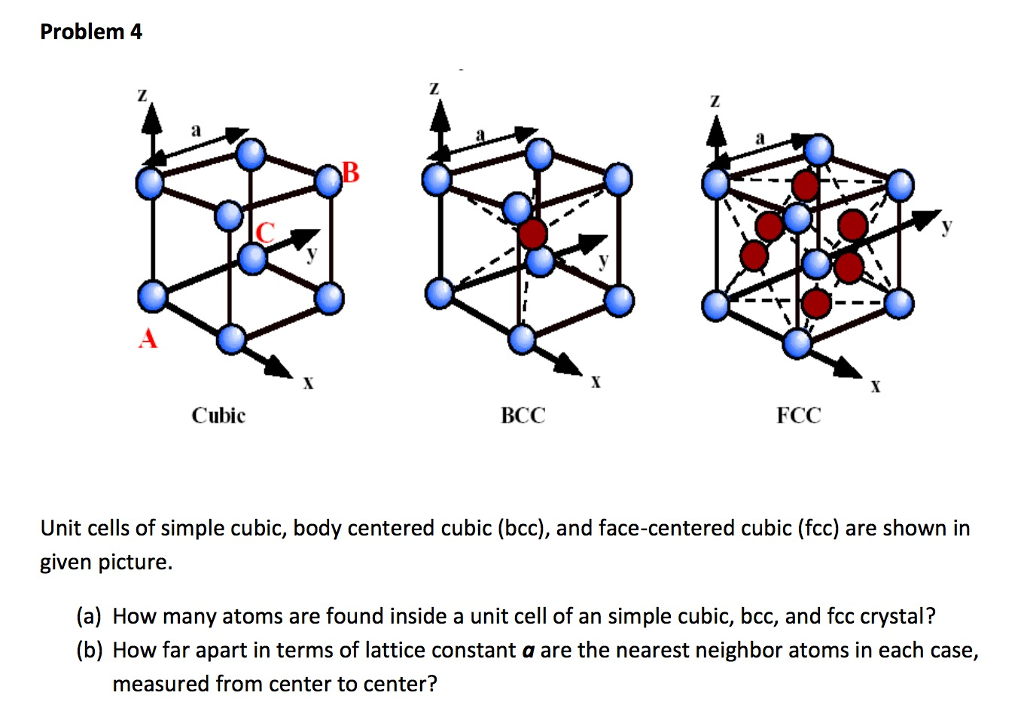
![Solved 3. Face Centered Cubic Structure [10 pts] Platinum is | Chegg.com Solved 3. Face Centered Cubic Structure [10 pts] Platinum is | Chegg.com](https://media.cheggcdn.com/media/1eb/1eb9fa27-1dc7-4453-878c-a7f39a79cbf4/phppeImhN.png)